On November 29, 2021, Ton-Bridge Medical's Flow Diverter has successfully completed the enrollment of the last patient in the national prospective multi-center single-arm clinical trial project. The clinical trial has achieved important phased progress in less than 5 months since the first case was enrolled in July 2021, once again demonstrating Ton-Bridge Medical's strong R&D strength and efficient execution.
The clinical trial was led by Changhai Hospital. The team of Professor Liu Jianmin from the Cerebrovascular Disease Center of the hospital served as the PI. Several authoritative clinical centers across the country, including Huashan Hospital affiliated to Fudan University, participated in the clinical trial.
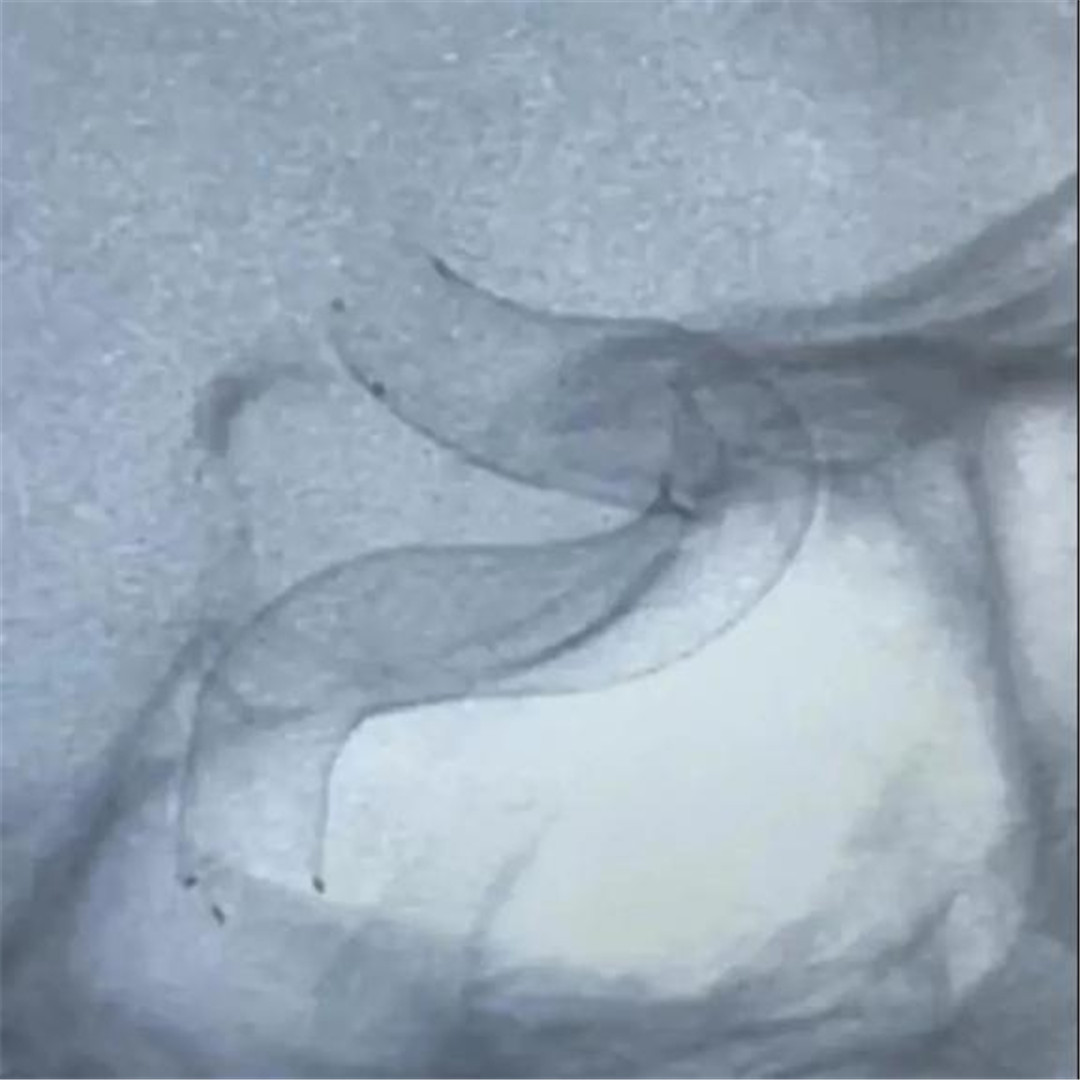
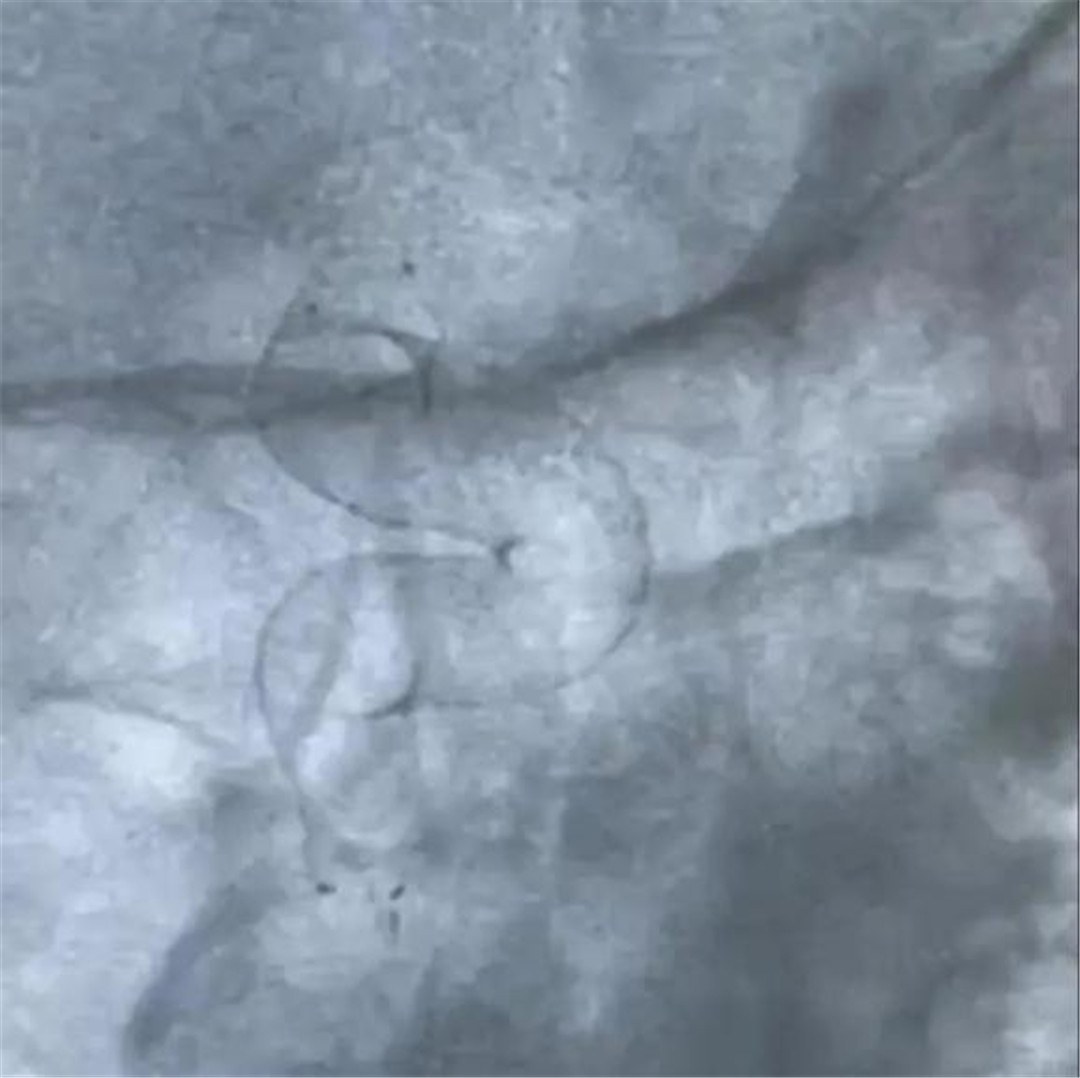
Following the efficacy and safety study of Ton-Bridge Medical's Flow Diverter for endovascular treatment of huge unruptured intracranial aneurysms, Ton-Bridge Medical carried out another research of effective and safe treatment focused on endovascular treatment of small unruptured aneurysms.
The Flow Diverter is a revolutionary product for the treatment of intracranial aneurysms, and has gradually become one of the important means for the treatment of various intracranial aneurysms in recent years. Ton-Bridge medical blood flow guiding device is made of nickel-titanium alloy material, which has the advantages of full imaging and good adherence. It can better isolate aneurysms and reconstruct diseased blood vessels, with high safety.
Several experts who participated in the clinical trial said that the product design of the Flow Diverter uses whole-body imaging materials, and has the characteristics of distal stent closure, bell-mouth design, moderate radial force, and strong adherence. It is expected that this highly innovative product will be launched as soon as possible to benefit patients.
Since 2021, the research and development and evidence collection of various products of Ton-Bridge Medical have continued to advance efficiently. In the future, Ton-Bridge Medical will continue to improve its competitiveness in product development and commercialization, and bring better and more inclusive medical devices and solutions to domestic patients and doctors.
Post time: Apr-19-2022
